The common material of solar mounting system is steel, so steel corrosion is the key consideration in the design of the support. This article starts with a simple principle of corrosion. The next article will discuss with you from the parts of "atmospheric corrosion", "soil corrosion" and "contact corrosion", and analyze and explain how to consider and avoid it in the design stage The impact of corrosion.
The fate of steel
Because steel has relatively stable physical and mechanical properties, good plasticity, and high strength, steel is the most widely used metal material in industrial buildings. However, steel solar mounting system are often easily corroded in nature. The lighter ones produce rust which affects the appearance, and the heavy ones destroy the structural quality, leading to the collapse of the building and causing accidents. It is said that one-tenth of the world's steel is scrapped due to corrosion. It can be seen that corrosion is an eternal topic that steel cannot avoid.
To apply the classic sentence from the American drama ``Game of Thrones'' is:
"AllMen(Metal) Must Die"
Nail experiment
When we studied oxidation-reduction reactions in high school, we often explained the corrosion of nails. Here, Xiaoshudong takes everyone to review this part of knowledge, in order to draw up the basic theory of steel corrosion.
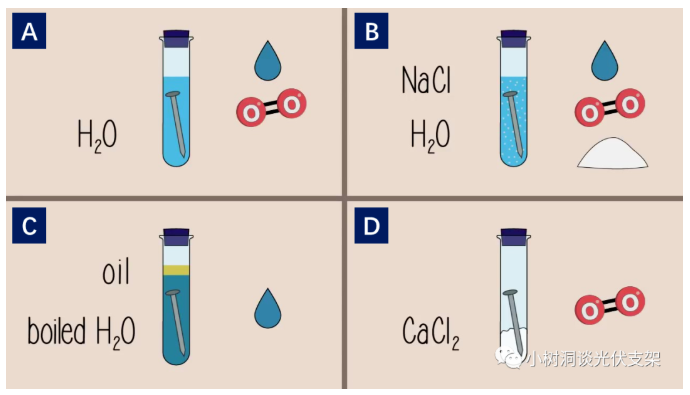
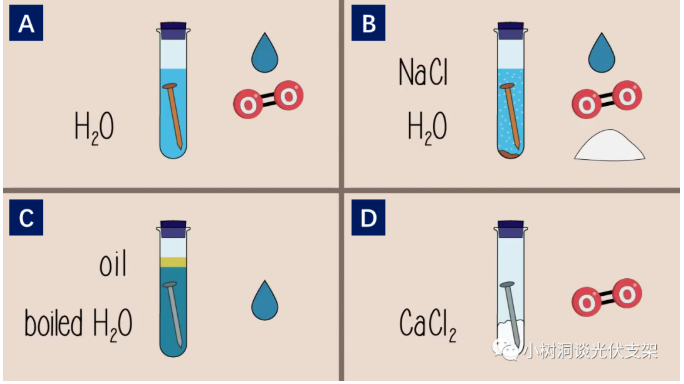
Group A nails: add water; water and oxygen
Group B iron nails: add water, sodium chloride; water, oxygen, chloride ions
Group C nails: add heated water and oil; only water
Group D iron nails: calcium chloride added; oxygen only
Group A is the reference group, and the other groups of experiments contain three elements: water, oxygen, and chloride ions. In order to compare the effects of these three elements on iron nails.
After a few days, the results of the experiment were obvious. Among them, only the nails in groups A and B rusted, and the rust in group B was more severe than that in group A. The C and D groups showed no signs of rust. From this experiment, the following conclusions can be obtained:
The comparison between group B and group A shows that chloride ion accelerates the corrosion of iron.
Group C compares group A, indicating that oxygen is a necessary condition for corrosion.
Group D compares group A, indicating that water is a necessary condition for corrosion.
Corrosion mechanism
According to the above experiments, we can have some perceptual understanding of the causes of corrosion, but to truly understand the mechanism of corrosion, it is necessary to conduct in-depth theoretical research on it. We observed that a new substance was produced after steel corrosion, and this change to produce new species originated from a certain chemical reaction.
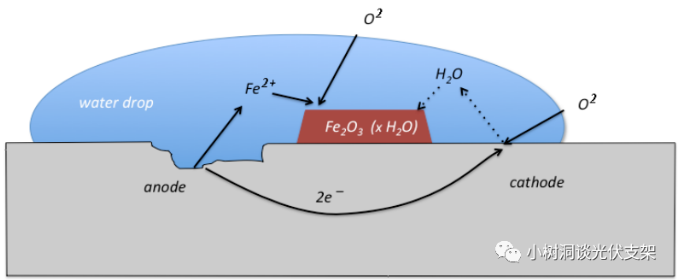
Iron, oxygen, and water, these three elements combine to form a "primary battery", and this kind of corrosion is also called "electrochemical corrosion". Its essence is a redox reaction, and the root cause is what we often call "electron migration".
After we understand the redox theory, we can quantitatively explain the corrosion mechanism:
Under the action of oxygen, iron atoms lose electrons to produce iron oxide.
Water acts as an electrolyte necessary for electron migration during the reaction.
The acid-base substances in nature act as catalysts to accelerate the reaction.
▵Iron corrosion legend
The principle of iron corrosion seems simple, but sometimes because everyone does not understand this phenomenon, it causes many regrettable accidents. The more famous one is the "Cangzhou Iron Lion".
▵Cangzhou Iron Lion: Also known as "Zhenhai Roar", it was built in Guangshun three years later (AD 935) and has a history of more than a thousand years. In the 1950s, according to the suggestion of the Soviet "experts", the Cangzhou Cultural Insurance Company built a small pavilion on top of the iron lion, hoping to slow down the further corrosion of the iron lion, but because the pavilion prevented the rapid evaporation of water vapor, the iron lion , Water, and oxygen in the pavilion constitute a "primary battery", which accelerates the corrosion rate of the iron lion. The pavilion was finally demolished after 20 years of construction.
For the design of solar mounting system, we mainly consider the corrosion effects from three aspects. They are:
▵Atmospheric corrosion: The upper support structure exposed in the atmosphere will interact with oxygen and water in the air to produce corrosion. This corrosion is the most common and the most easily noticed.
▵Soil corrosion: The steel pile foundation buried deep underground will undergo more complex chemical reactions under the action of various substances in the soil, resulting in corrosion. Because of the long-term presence in the soil, this kind of corrosion is difficult to detect.
▵Contact corrosion: Between different metals, due to the difference in potential, electron gains and losses will occur, resulting in electrochemical corrosion, which is often ignored by designers.
In the following series, Xiaoshudong will briefly explain how to quantitatively consider and calculate the above three corrosion conditions at the initial stage of the design.
Anti-corrosion method
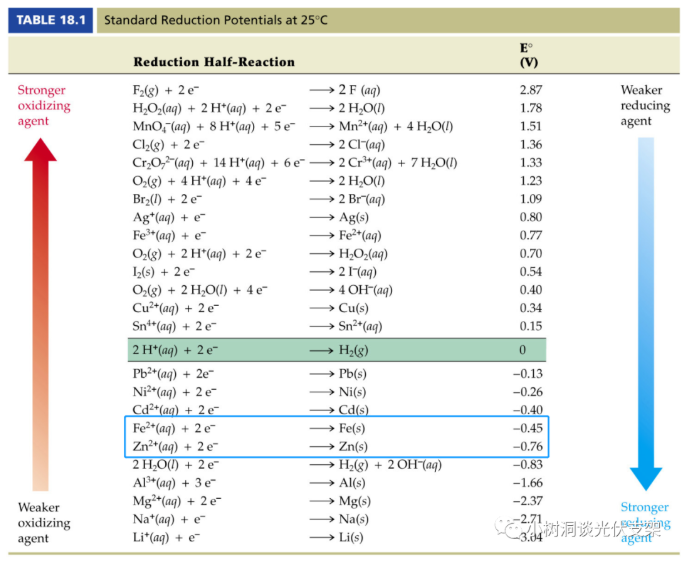
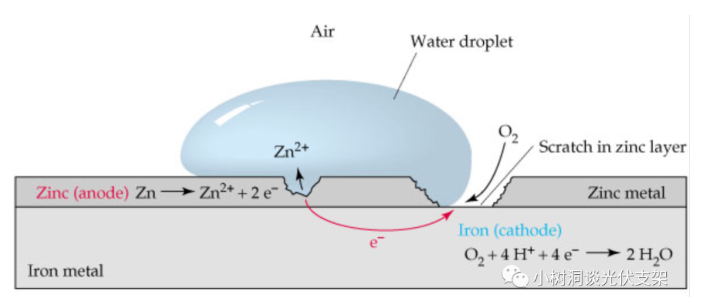
In order to slow down or even avoid steel corrosion, a common method in the industry is to "galvanize" the surface of the steel. The standard potential of zinc at 25°C is -0.76V, which is lower than the -0.45V of iron. Therefore, zinc is "active" "Strong than iron. Zinc and iron form a "primary battery" in a humid environment. Like all electrochemical corrosion principles, this combination of primary batteries is:
"Sacrificial anode (zinc), protective cathode (iron)"
▵Standard electric potential of each element
The lower the rank, the easier it is to lose electrons
Due to the galvanic effect formed by zinc and iron, even if the zinc coating is slightly damaged during transportation or installation, it will not affect the corrosion resistance of the zinc coating. Of course, extra care must be taken for deeper damage, and zinc supplementation is required for these areas.
▵A galvanic battery formed by zinc, iron and oxygen in a humid environment
Even if the galvanized layer is slightly scratched
Will not affect its corrosion ability
In addition, zinc reacts with air and water to produce a new product: basic zinc carbonate. This substance has a dense surface and is not easy to dissolve in water. It can well isolate air and iron, thus forming again A layer of "physical protection" is added to further slow down the corrosion of steel.
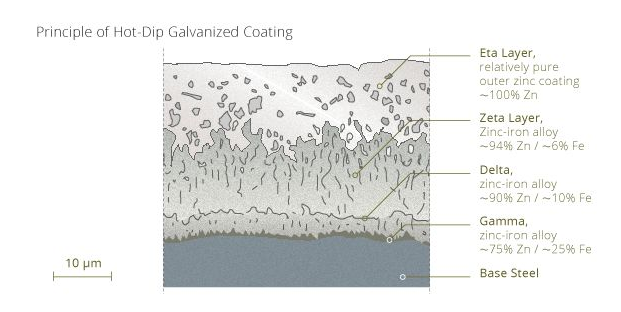
Common galvanizing processes are: hot-dip galvanizing, pre-galvanizing, and electro-galvanizing (cold galvanizing). The method and steps of galvanizing are not described here too much. Of course, regardless of the galvanizing process, the zinc layer itself is also composed of many different materials, such as the hot-dip galvanizing layer in the following figure:
In addition, there are anti-corrosion processes such as magnesium aluminum zinc plating, VCI, epoxy coating and weathering steel without any surface treatment.

Above news from CHIKO Sales & Marketing department
Add: No 680 Xingwen Rd. Jiading dist. Shanghai 201801 China Email: Chiko@Chikolar.com
Website: http://www.chikolar.com Fax : +86 (21)59972938
Phone: +86(21) 59972267 59973712 59973713--811